|
6/14/2023 0 Comments Negatively charged atomBecause the reduction in mass upon the formation of an atom is small, the mass number is usually the same as the atomic weight rounded to the nearest integer (for example, the atomic weight of chlorine-37 is 36.966, which is rounded to 37). The negatively charged electrons follow a random pattern. Ions with a positive charge are called cations. 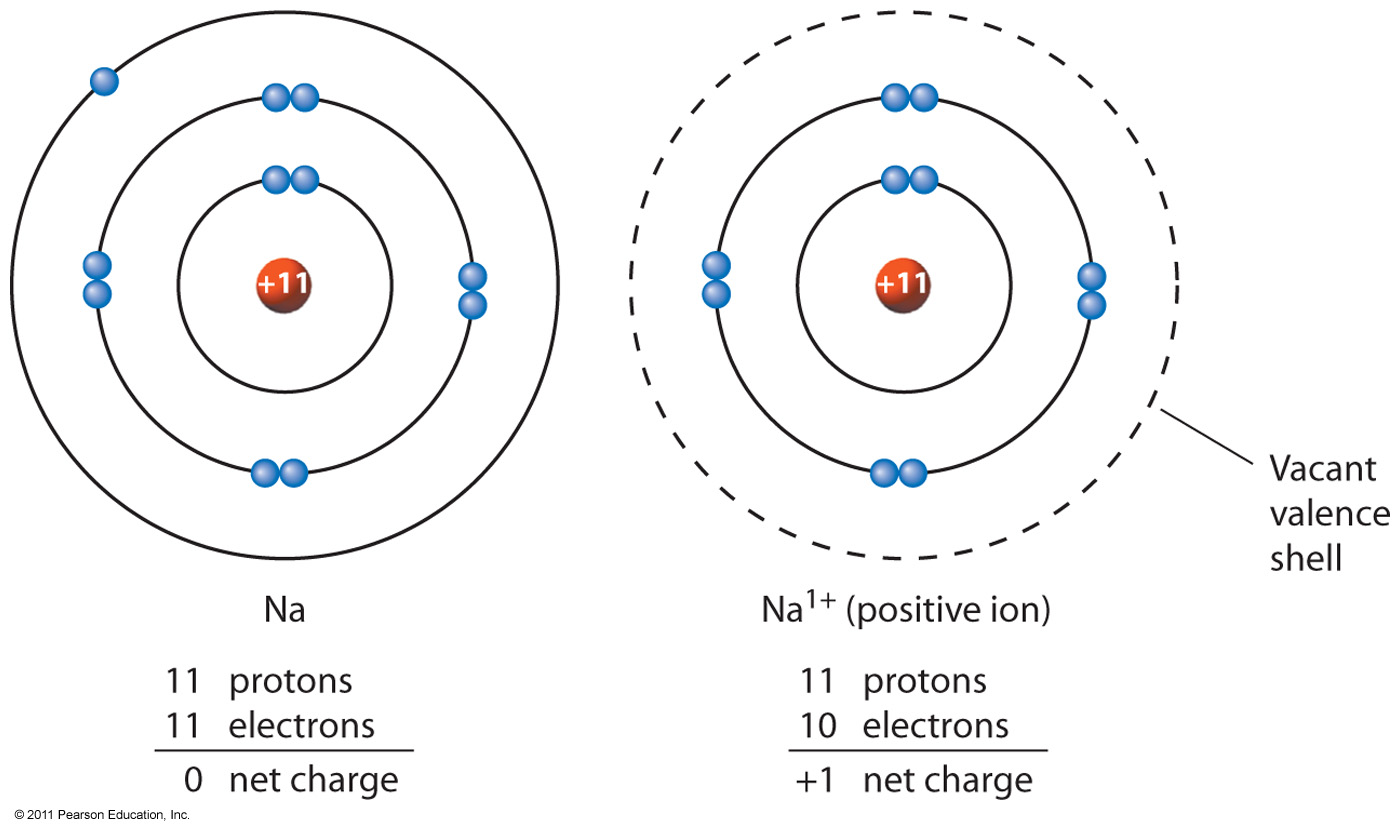
The net charge of an ion is not zero because its total number of electrons is unequal to its total number of protons. An ion is an atom or group of atoms that has an electric charge. When protons, neutrons, and electrons combine to form an atom, some of their mass is converted to energy and is given off (this is the source of energy in nuclear fusion reactions because the atom cannot be broken down into its fundamental particles unless the energy for the missing mass is supplied from outside it, this energy is called the binding energy of the nucleus).Įach isotope of an element is characterized by an atomic number (total number of protons), a mass number (total number of protons and neutrons), and an atomic weight (mass of atom in atomic mass units). Thomson discovered the existence of the electron, marking the beginning of modern atomic physics. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by convention. 
The total mass of an atom is called its atomic weight, and this is approximately the sum of the masses of its constituent protons, neutrons, and electrons. For instance, a chlorine atom gains one electron to form a chloride ion, Cl, which is an anion. An anion is devised by an atom’s gain of one or more electrons. Within a group (family) of elements, atoms form ions of a certain charge.\) (or 238U). The first is the tiny atomic nucleus, which is in the center of the atom and contains positively charged particles called protons and neutral, uncharged, particles called neutrons. An anion or a negatively charged ion: Fluoride ions, F, or sulfide ion, S², are called anions because they are negatively charged ions. An atom of an element is most stable when its outer electron shell is completely filled or half-filled. The charge on an atom is related to its valence electrons or oxidation state. You can use this chart to predict whether or not an atom can bond with another atom. The atom becomes negatively charged., The Venn diagram compares protons with neutrons. This is a chart of the most common charges for atoms of the chemical elements. Cations vs anions what’s the difference Cations are ions that are positively charged. 
However, if they are not balanced, they will be charged. The oxygen atom is slightly negatively charged, and the carbon and hydrogen atoms are slightly positively charged. 4 minutes If an atom, or atoms, has a balanced number of electrons (negative charge) and protons (positive charge) they are neutral overall. 
The charge that an atom acquires when it becomes an ion is related to the structure of the periodic table. Study with Quizlet and memorize flashcards containing terms like How does an atom change if all of its electrons are removed The atoms volume increases. An oxygen atom is much more electronegative than carbon or hydrogen atoms, so both carbon-oxygen and hydrogen-oxygen bonds are polar. Under normal circumstances, the electrons cannot escape from the atom since positive and negative electrical charges attract each other and therefore.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
